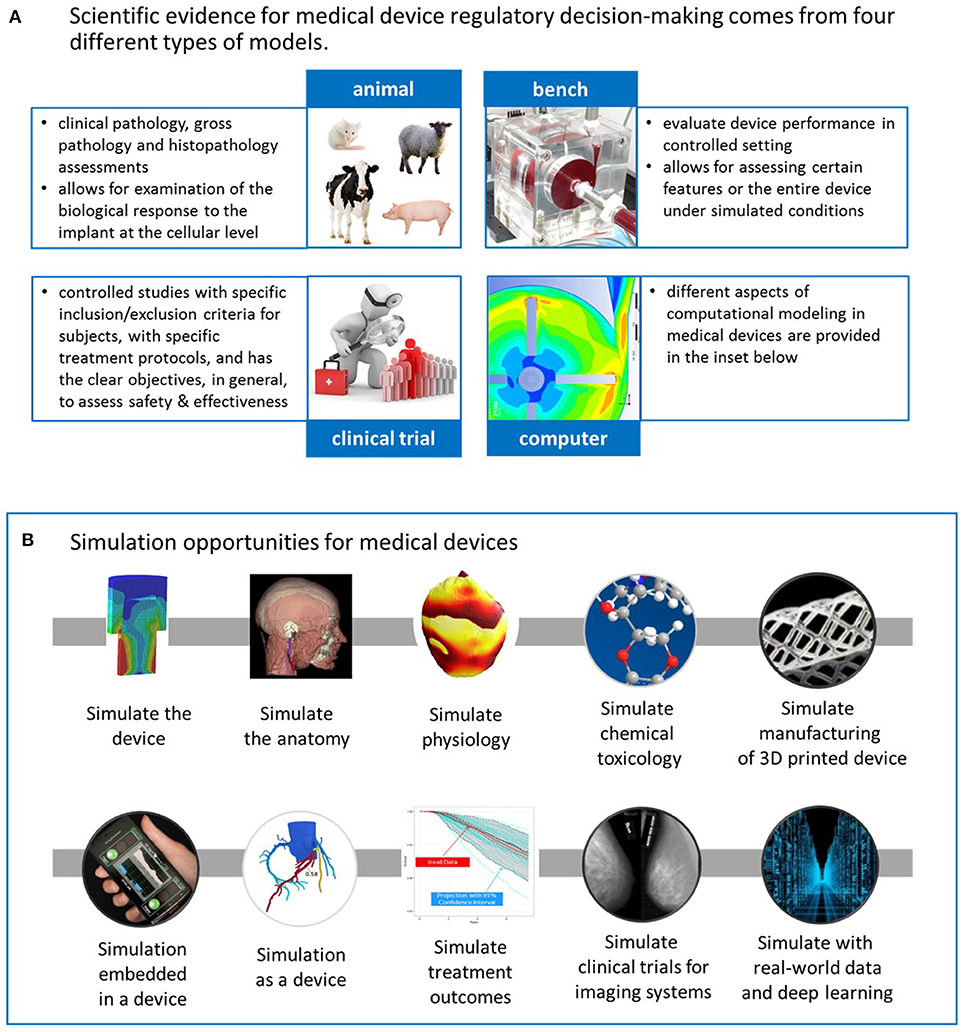
Frontiers | Advancing Regulatory Science With Computational Modeling for Medical Devices at the FDA's Office of Science and Engineering Laboratories

Continuous Glucose Monitors and Automated Insulin Dosing Systems in the Hospital Consensus Guideline - Rodolfo J. Galindo, Guillermo E. Umpierrez, Robert J. Rushakoff, Ananda Basu, Suzanne Lohnes, James H. Nichols, Elias K.
Navigating the Regulatory Pathway for Medical Devices—a Conversation with the FDA, Clinicians, Researchers, and Industry Experts | SpringerLink

Insulin Lispro with Continuous Subcutaneous Insulin Infusion is Safe and Effective in Patients With Type 2 Diabetes: A Randomized Crossover Trial of Insulin Lispro Versus Insulin Aspart* - Endocrine Practice
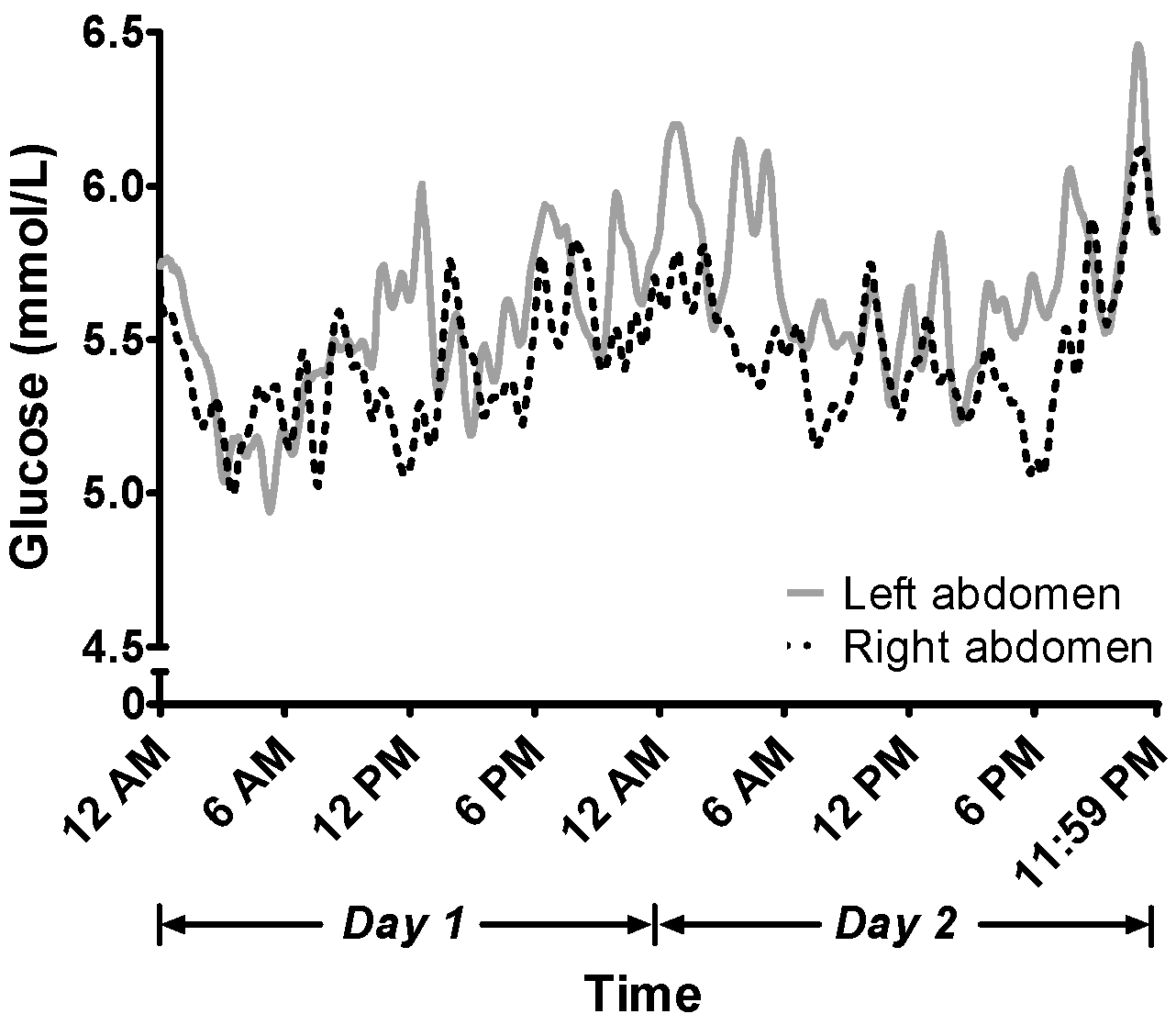
Biosensors | Free Full-Text | Consistency of Continuous Ambulatory Interstitial Glucose Monitoring Sensors
![Selecting the Appropriate Continuous Glucose Monitoring System – a Practical Approach | [current-page:pager]touchENDOCRINOLOGY Selecting the Appropriate Continuous Glucose Monitoring System – a Practical Approach | [current-page:pager]touchENDOCRINOLOGY](https://www.touchendocrinology.com/wp-content/uploads/sites/5/2018/02/table1-summary-of-char.png)
Selecting the Appropriate Continuous Glucose Monitoring System – a Practical Approach | [current-page:pager]touchENDOCRINOLOGY

Reference Guide for Integrating Continuous Glucose Monitoring Into Clinical Practice - Davida F. Kruger, Steve V. Edelman, Deborah A. Hinnen, Christopher G. Parkin, 2019
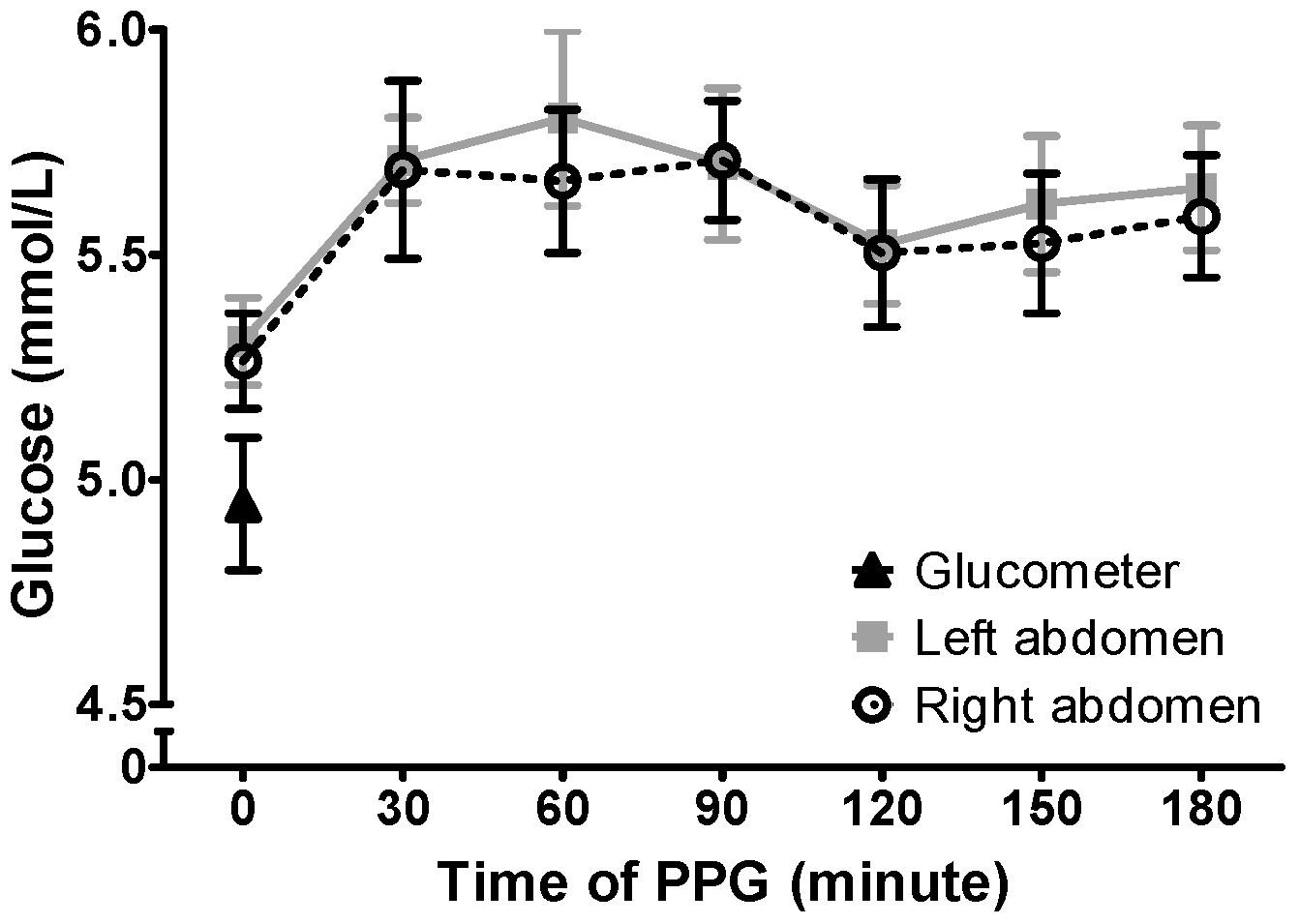
Biosensors | Free Full-Text | Consistency of Continuous Ambulatory Interstitial Glucose Monitoring Sensors

Medical devices, smart drug delivery, wearables and technology for the treatment of Diabetes Mellitus - ScienceDirect

PDF) Adverse event using Medtronic NIM™ EMG endotracheal tube on a patient receiving anesthesia for hemithyroidectomy: a case report

Monitoring Technologies- Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control - Endotext - NCBI Bookshelf
This policy addresses policy and coverage guidelines for Implantable and Non-Implantable Continuous Glucose Monitors (CGM) Home
P160017/S031: FDA Summary of Safety and Effectiveness Data Page 1 SUMMARY OF SAFETY AND EFFECTIVENESS DATA (SSED) I. GENERAL IN